UNIT IV: Micro-propagation & Advanced Techniques
Micro-propagation - principles and concepts, commercial exploitation in horticultural crops. Techniques - in vitro clonal propagation, direct organogenesis, embryogenesis, micrografting, meristem culture. Hardening, packing and transport of micro-propagules.
Table of Contents
Micro-propagation – Principles, Concepts & Commercial Exploitation in Horticultural Crops
1. INTRODUCTION
Micro-propagation = In vitro (lab-based) clonal propagation of plants using small plant parts (explants) under sterile and controlled conditions.
👉 It is a major application of plant tissue culture.
1.2 Key Concept
Based on Totipotency → Every plant cell has the ability to regenerate into a whole plant
1.3 Importance
- Rapid multiplication
- Disease-free planting material
- Year-round production
- Uniform plants
2. PRINCIPLES OF MICRO-PROPAGATION
2.1 Totipotency
- Single cell → whole plant
- Basis of all tissue culture techniques
2.2 Aseptic Conditions
- Sterile environment is essential
- Prevents contamination by: Bacteria, Fungi
2.3 Controlled Environment
- Temperature: 22–27°C
- Light: 1000–3000 lux
- Photoperiod: 16 hours light / 8 hours dark
2.4 Nutrient Medium
Most commonly used medium: 👉 Murashige and Skoog medium
- Components:
- Macronutrients (N, P, K, Ca, Mg)
- Micronutrients (Fe, Zn, Mn)
- Vitamins
- Carbon source (Sucrose: 2–3%)
- Growth regulators
2.5 Role of Plant Growth Regulators (PGRs)
| Hormone | Function |
|---|---|
| Auxins (IAA, IBA, NAA) | Root formation |
| Cytokinins (BAP, Kinetin) | Shoot multiplication |
| Gibberellins | Shoot elongation |
👉 Auxin : Cytokinin ratio controls organ formation
3. CONCEPTS OF MICRO-PROPAGATION
Small plant part used to initiate culture: Shoot tip, Meristem, Leaf, Node.
3.2 Stages of Micro-propagation
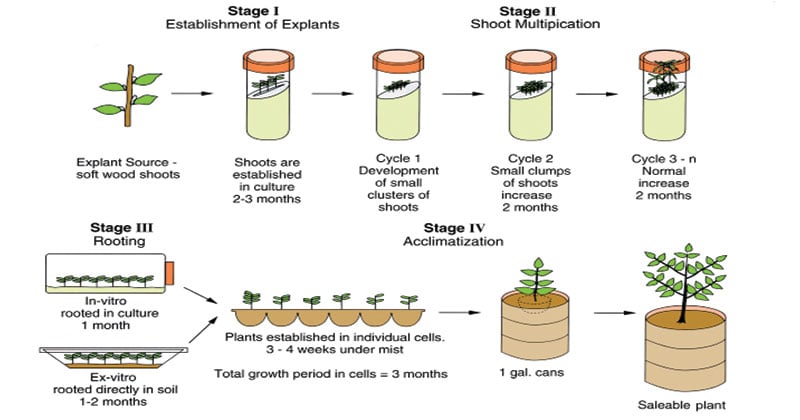
- Stage I: Establishment
- Selection of explant
- Surface sterilization: HgCl2 (0.1%) or NaOCl
- Inoculation on culture medium
- Stage II: Multiplication
- Rapid shoot multiplication
- Use of cytokinins (BAP)
- Subculturing every 3–4 weeks
- Stage III: Rooting
- Transfer shoots to rooting medium
- Auxins used (IBA, NAA)
- Stage IV: Hardening (Acclimatization)
- Transfer to soil/soilless media
- Gradual exposure to external environment
- High humidity initially (80–90%)
4. TYPES OF MICRO-PROPAGATION
- Meristem Culture: Used for virus-free plants. Very small shoot tip (0.1–0.5 mm).
- Callus Culture: Undifferentiated mass of cells. Can regenerate plants.
- Somatic Embryogenesis: Embryo formation from somatic cells. Important for large-scale propagation.
- Organogenesis: Direct formation of shoots/roots from explants.
5. COMMERCIAL EXPLOITATION IN HORTICULTURAL CROPS
5.1 Advantages for Commercial Use
- Rapid multiplication (1000s plants/year)
- Disease-free (virus-free) plants
- Uniform growth and yield
- Space-saving
- Export-quality planting material
5.2 Major Crops Propagated
- Fruit Crops: Banana, Strawberry, Pineapple, Apple
- Plantation Crops: Tea, Coffee, Oil palm
- Ornamentals: Orchid, Chrysanthemum, Gerbera
- Vegetable Crops: Potato (seed production)
5.3 Commercial Process Flow
Selection of elite plant → Establishment of culture → Mass multiplication → Rooting → Hardening → Nursery raising → Field planting
5.4 Industrial Scale Production
- Done in tissue culture labs
- Automated systems: Bioreactors, Controlled environment chambers
5.5 Role of Institutions
- ICAR → Research & protocols
- National Horticulture Board → Promotion & subsidy
- Private tissue culture labs → commercial supply
6. LIMITATIONS
- High initial cost
- Requires skilled labour
- Risk of contamination
- Somaclonal variation (genetic variation)
Techniques: In vitro Clonal Propagation, Direct Organogenesis, Somatic Embryogenesis
1. IN VITRO CLONAL PROPAGATION (MICROPROPAGATION)
1.1 Definition: Production of genetically identical plants (clones) using tissue culture under sterile conditions.
1.2 Principle: Based on Totipotency. Controlled use of growth regulators → shoot & root formation.
1.3 Steps
- Explant selection (shoot tip/node)
- Surface sterilization
- Inoculation on medium
- Shoot multiplication
- Rooting
- Hardening
1.4 Key Features
- Rapid multiplication (10–1000 plants/explant/year)
- Uniform plants
- Used in banana, potato, ornamentals
2. DIRECT ORGANOGENESIS
2.1 Definition: Formation of shoots or roots directly from explant without callus stage.

2.2 Principle: Cells differentiate directly into organs. Avoids genetic variation.
2.3 Process
- Explant placed on medium
- Cytokinin induces shoot formation
- Shoots elongated
- Rooting induced
2.4 Characteristics
- Faster than indirect method
- Genetically stable
- High success in nodal cultures
2.5 Applications
- Micropropagation of fruit crops
- Rapid multiplication of elite genotypes
3. SOMATIC EMBRYOGENESIS
3.1 Definition: Formation of embryos from somatic (non-reproductive) cells.
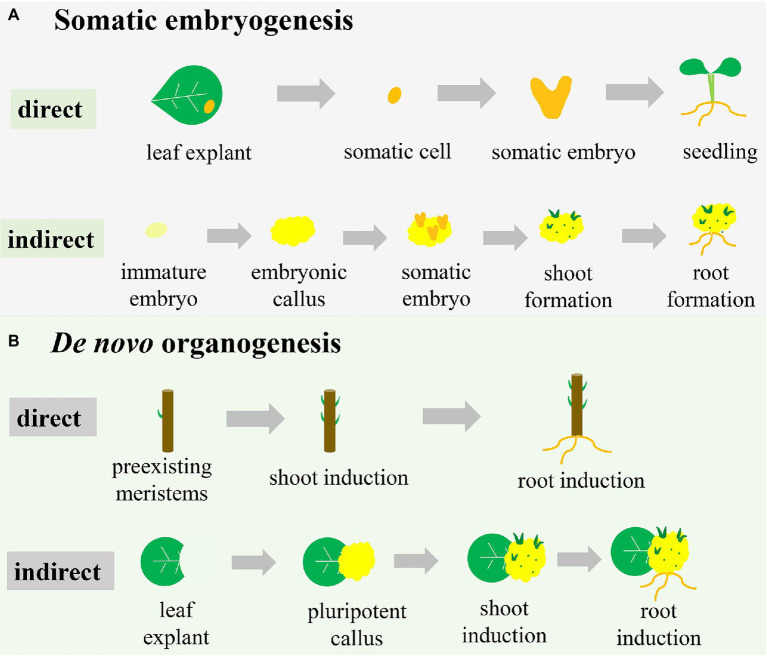
3.2 Principle: Somatic cells behave like zygotic embryos. Develop into complete plants.
3.3 Types
- Direct embryogenesis (without callus)
- Indirect embryogenesis (via callus)
3.4 Stages
- Induction → callus formation
- Embryo formation: Globular → Heart → Torpedo
- Maturation
- Germination → plantlet
3.5 Advantages
- Large-scale propagation
- Synthetic seed production
- Automation possible
3.6 Applications
Banana, Oil palm, Coffee
4. MICROGRAFTING & MERISTEM CULTURE
4. MICROGRAFTING
4.1 Definition: Grafting of very small scion (meristem/shoot tip) onto a seedling rootstock under in vitro conditions.
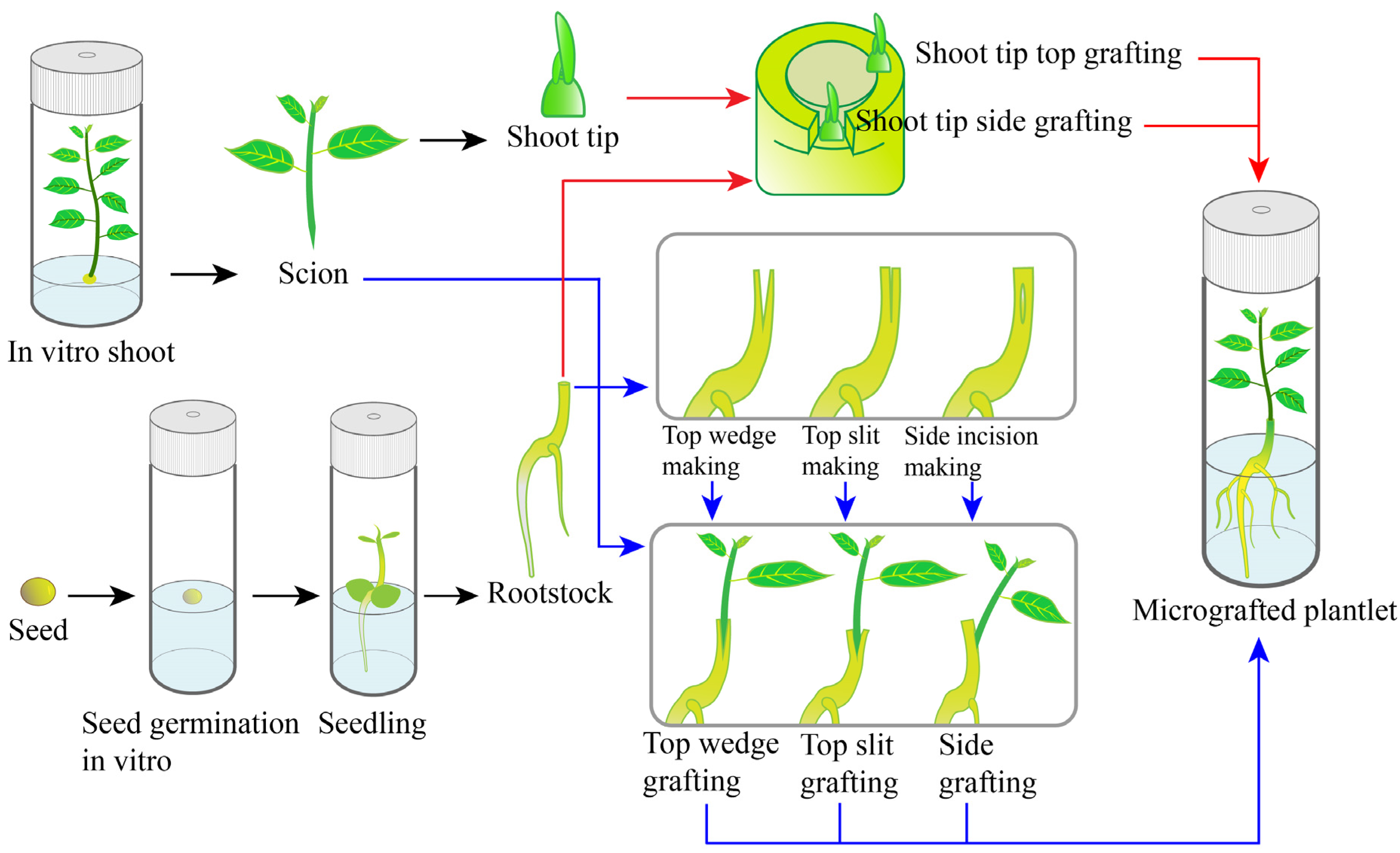
4.2 Principle: Combines Tissue culture + Conventional grafting. Used for disease elimination.
4.3 Process
- Rootstock grown in vitro
- Tiny shoot tip excised
- Grafted onto rootstock
- Cultured in sterile conditions
4.4 Applications
Citrus (very important), Apple, Grapes.
4.5 Advantages
- Produces virus-free plants
- Rapid multiplication
- Overcomes incompatibility
5. MERISTEM CULTURE
5.1 Definition: Culture of apical meristem (0.1–0.5 mm) to produce disease-free plants.

5.2 Principle: Meristem is free from viruses. Viruses do not easily invade actively dividing cells.
5.3 Process
- Excise meristem under microscope
- Culture on nutrient medium
- Shoot development
- Rooting
- Hardening
5.4 Applications
Potato (virus-free seed), Banana, Strawberry.
5.5 Advantages
- Virus-free plants
- Maintains genetic purity
6. COMPARISON OF TECHNIQUES
| Technique | Key Feature | Advantage | Limitation |
|---|---|---|---|
| Clonal propagation | Mass multiplication | Uniform plants | Costly |
| Direct organogenesis | No callus | Stable | Limited explants |
| Somatic embryogenesis | Embryo formation | Large scale | Variation possible |
| Micrografting | Tiny graft | Virus-free | Skill required |
| Meristem culture | Shoot tip culture | Disease-free | Slow initial growth |
Hardening, Packing and Transport of Micro-propagules
1. INTRODUCTION
• Delicate
• Non-functional cuticle
• Poor stomatal control
• Weak root system
👉 Therefore, hardening (acclimatization) is essential before field transfer.
2. HARDENING (ACCLIMATIZATION)
2.1 Definition: Gradual adaptation of in vitro plantlets to external (ex vitro) conditions.
2.2 Need for Hardening
In vitro plantlets have:
- Thin cuticle → high water loss
- Non-functional stomata
- Low photosynthetic capacity
- High humidity adaptation
👉 Sudden transfer → high mortality
2.3 Types of Hardening
- Primary Hardening: Done in Growth chamber, Mist chamber, or Polyhouse. 👉 Controlled conditions.
- Secondary Hardening: Done in Nursery (shade net house). 👉 Gradual exposure to natural conditions.
2.4 Steps in Hardening Process
- Step 1: Removal from Culture: Remove plantlets from culture vessels. Wash roots to remove agar medium.
- Step 2: Fungicide Treatment: Dip in fungicide (Carbendazim 0.1% or similar). 👉 Prevent fungal infection.
- Step 3: Transplanting to Medium: Plant in sterilized medium (Cocopeat, Vermiculite, Perlite). 👉 Ideal mixture: Cocopeat : Vermiculite = 1:1.
- Step 4: High Humidity Maintenance: Maintain 80–90% RH initially. Use Mist chamber or Fogging system.
- Step 5: Gradual Exposure: Reduce humidity gradually. Increase light intensity stepwise. Open vents slowly.
- Step 6: Nutrient Supply: Foliar spray of dilute nutrient solution (e.g., ½ MS). Helps growth establishment.
2.5 Environmental Conditions
| Factor | Ideal Range |
|---|---|
| Temperature | 25–30°C |
| Humidity | 80–90% (initial) |
| Light | Low → moderate |
| Shade | 50–75% shade net |
2.6 Duration & Importance
- Primary hardening: 2–4 weeks | Secondary hardening: 4–6 weeks
- Improves survival rate (up to 80–95%)
- Develops functional: Roots, Stomata, Photosynthetic system
3. PACKING OF MICRO-PROPAGULES
3.1 Objectives
- Prevent mechanical damage, moisture loss, and contamination.
- Maintain plant health during transport.
3.2 Packing Methods
- Polybag Packing: Plantlets packed in polybags with growing medium. Most common method.
- Tray Packing: Plug trays used for bulk transport. Suitable for short distances.
- Moist Packing: Roots wrapped in moist sphagnum moss or tissue paper.
- Box Packaging: Corrugated boxes with ventilation holes and cushioning material.
3.3 Precautions
Avoid overpacking. Maintain moisture (not waterlogged). Labeling of varieties. Use ventilated packaging.
4. TRANSPORT OF MICRO-PROPAGULES
4.1 Objectives & 4.2 Key Requirements
- Deliver plants safely, maintaining physiological condition.
- Temperature Control: Maintain 15–25°C. Avoid heat stress.
- Humidity Maintenance: Prevent desiccation. Use moist packing.
- Ventilation: Prevent CO2 accumulation. Avoid fungal growth.
- Handling: Gentle handling. Avoid shaking and damage.
4.3 Modes of Transport & 4.4 Post-Transport Care
- Road (short distance) or Air transport (export material).
- Immediate unpacking, light irrigation, and keep in shade initially.
5. PROBLEMS DURING HARDENING & TRANSPORT
- High Mortality: Due to sudden environmental change
- Desiccation: Poor humidity control
- Fungal Infection: Poor sanitation
- Mechanical Damage: Improper packing
7. IMPORTANT EXAM POINTS (REVISION)
- Micro-propagation based on totipotency
- MS medium is most widely used
- 4 stages: Establishment, Multiplication, Rooting, Hardening
- Cytokinin → shoot
- Auxin → root
- Widely used in banana, potato, ornamentals
- Micropropagation → cloning via tissue culture
- Direct organogenesis → no callus stage
- Somatic embryogenesis → embryo from somatic cells
- Micrografting → in vitro grafting
- Meristem culture → virus-free plants
- Hardening = adaptation from in vitro → ex vitro
- Two stages: Primary (controlled), Secondary (nursery)
- RH initially 80–90%
- Cocopeat + vermiculite commonly used
- Packing → prevent moisture loss & damage
- Transport → maintain temperature & humidity
8. SOURCES
- Hartmann & Kester – Plant Propagation: Principles and Practices
- Bhojwani & Razdan – Plant Tissue Culture: Theory and Practice
- ICAR – Handbook of Horticulture
- NHB Guidelines – National Horticulture Board
- ICAR Tissue Culture Manuals – ICAR
- ICAR Protocols – ICAR
- FAO Tissue Culture Manuals
